Prof. Zach Adam
The Proteolytic Machinery of Chloroplasts
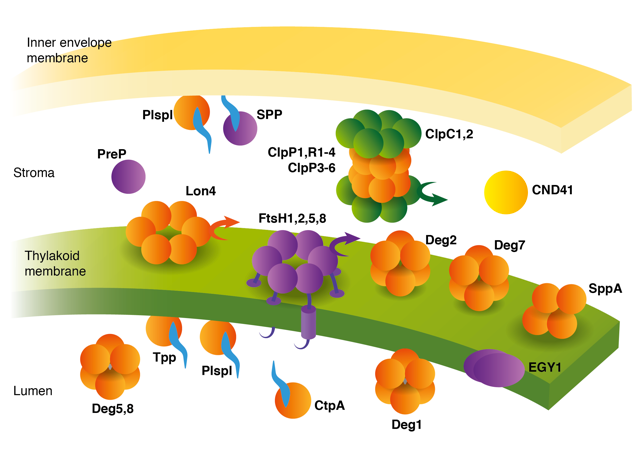
Proteolytic processes are involved in a wide range of functions throughout the life cycle of any cell. Chloroplast proteases thus affect photosynthesis at several different levels. The development of proplastids and etioplasts into chloroplasts involves not only the synthesis of a new set of proteins, but also massive degradation of the previous population. Limited proteolytic processing, for removal of targeting sequences, is an essential step in the import and sorting of nuclear-encoded chloroplast proteins, and in the maturation of some chloroplast-encoded proteins. Proteases participate in the biogenesis of some protein complexes, and in the recycling of proteins into free amino acids during senescence. Perhaps the most explored aspect of protein degradation in chloroplasts is its role in the response to changing environmental conditions. Increasing light intensities lead to photodamage, the repair of which requires accelerated removal of damaged proteins. Similarly, heat-denatured proteins that fail to refold properly are degraded.
Starting our lab in the 90’s, we have combined biochemical, molecular and genetic approaches, to identify and characterize the biochemical and physiological roles of a number of chloroplast proteases in all compartments of the organelle, with special emphasis on the thylakoid FtsH and Deg families, unraveling their role in the biogenesis and maintenance of the photosynthetic apparatus.


